|
12/17/2023 0 Comments Atomic radius definition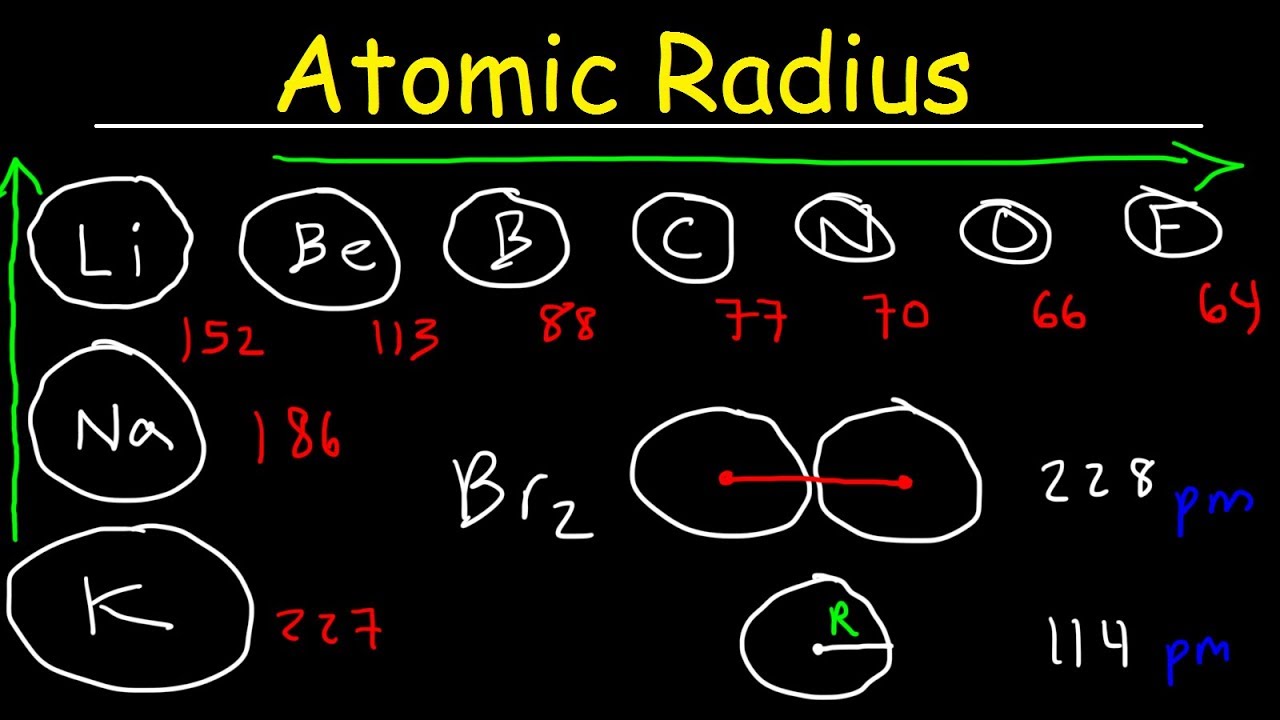 
The radius increases sharply between the noble gas at the end of each period and the alkali metal at the beginning of the next period. For instance, the radii generally decrease along each period (row) of the table, from the alkali metals to the noble gases and increase down each group (column). This is only a crude approximation, but it can provide quantitative explanations and predictions for many phenomena, such as the density of liquids and solids, the diffusion of fluids through molecular sieves, the arrangement of atoms and ions in crystals, and the size and shape of molecules.Ītomic radii vary in a predictable and explicable manner across the periodic table. Therefore, the radius of an atom is more than 10,000 times the radius of its nucleus (1–10 fm), and less than 1/1000 of the wavelength of visible light (400–700 nm).įor many purposes, atoms can be modeled as spheres. Moreover, in condensed matter and molecules, the electron clouds of the atoms usually overlap to some extent, and some of the electrons may roam over a large region encompassing two or more atoms.ĭespite these conceptual difficulties, under most definitions the radii of isolated neutral atoms range between 30 and 300 pm (trillionths of a meter), or between 0.3 and 3 angstroms. Rather, their positions must be described as probability distributions that taper off gradually as one moves away from the nucleus, without a sharp cutoff. The concept is difficult to define because the electrons do not have definite orbits, or sharply defined ranges. Under some definitions, the value of the radius may depend on the atom's state and context. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius.ĭepending on the definition, the term may apply only to isolated atoms, or also to atoms in condensed matter, covalently bound in molecules, or in ionized and excited states and its value may be obtained through experimental measurements, or computed from theoretical models. The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the nucleus to the boundary of the surrounding cloud of electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
